From Health Lab: Study defines key driver of aggressive ovarian cancer
Story originally posted on Michigan Medicine Health Lab, written by Nicole Fawcett.
Study defines key driver of aggressive ovarian cancer
Researchers created a mouse model to understand how the gene CDK12 drives high-grade serous tubo-ovarian carcinoma and identify potential treatment approaches
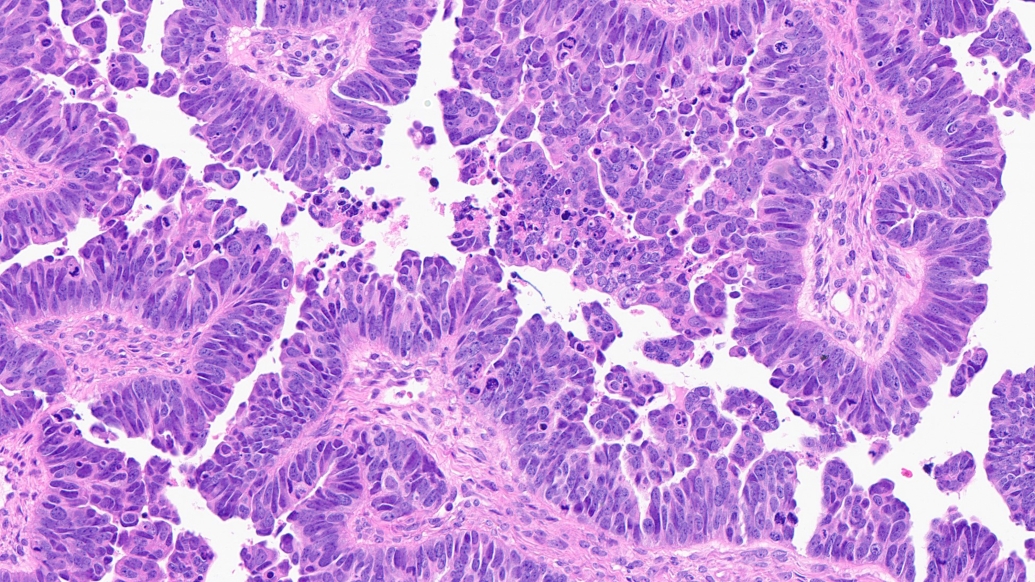
A study explains the genetic underpinnings of a rare and aggressive form of ovarian cancer – and offers a potential pathway for new treatments.
High-grade serous carcinoma, the most common type of ovarian cancer, usually begins in the fallopian tube, before spreading to the ovaries and other pelvic organs
